New Hope for Dry Macular Degeneration?
&srotate=0)
In the last twenty years, we have seen a revolutionary progression of treatments for “wet” age-related macular degeneration (ARMD), primarily injections of medications which act against vascular endothelial growth factor. Patients with sight-threatening vessel leakage and bleeding now have their disease stabilized and often improved through injections performed every 4 to 12 weeks or so.
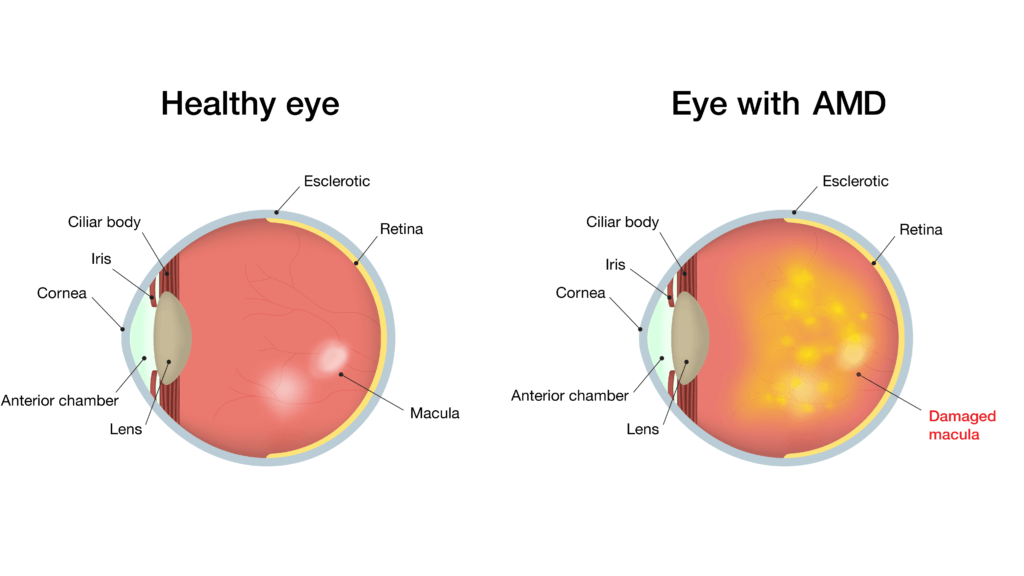
Most patients with age-related macular degeneration, however, have the “dry” form, which is less likely to bleed or leak. Over time, dry ARMD can transform into the wet form, so vigilance in monitoring is critical.
Fortunately, severe vision loss is less common in dry ARMD, and sight changes, when they occur, usually take many years. Unfortunately, when vision is lost to dry ARMD, the changes are generally irreversible. The medications used for wet ARMD are often not useful for patients with advanced dry ARMD.
How does dry ARMD lead to vision loss? A simple partial answer is: oxidative stress, which leads to inflammation, increased vessel permeability, and eventually cell death and neuronal degeneration.
A new treatment for intermediate and advanced dry ARMD is on the horizon, however. Recent research on risuteganib (Luminate, Allegro Ophthalmics) supports a significant anti-inflammatory effect on patients with dry ARMD, compared to sham injections.
The research showed a significant vision gain in dry ARMD patients twelve weeks after injection with risuteganib compared to controls.
Risuteganib is a small synthetic peptide which down-regulates proteins involved in cell-to-cell interactions called integrins, with a beneficial effect on oxidative stress. Risuteganib is also being tried for patients with diabetic macular edema.
The recently reported phase II risuteganib trial for dry ARMD only involved 40 patients, so we’re a long way off from knowing how useful the treatment really is. Also, because in its current formulation, risuteganib doesn’t stay active for very long periods, repeat injections would presumably be needed.
Benjamin H. Ticho, MD